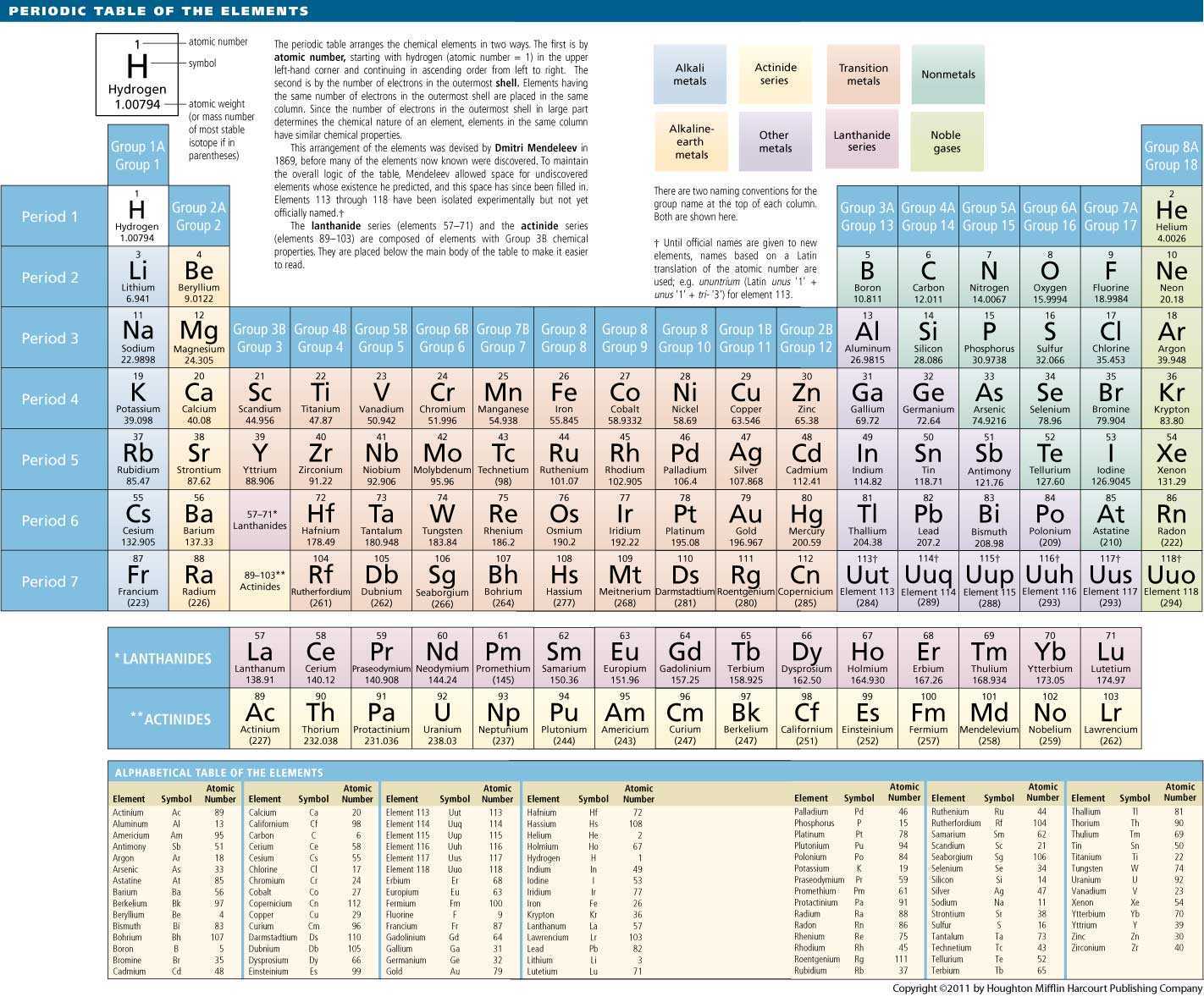
Elements that exhibit similar chemistry appear in vertical columns called groups (numbered 1–18 from left to right) the seven horizontal rows are called periods. It arranges of the elements in order of increasing atomic number. 
The periodic table is used as a predictive tool. As expected, semimetals exhibit properties intermediate between metals and nonmetals. Most solid nonmetals are brittle, so they break into small pieces when hit with a hammer or pulled into a wire. Nonmetals can be gases (such as chlorine), liquids (such as bromine), or solids (such as iodine) at room temperature and pressure. Nonmetals, in contrast, are generally poor conductors of heat and electricity and are not lustrous. Of the metals, only mercury is a liquid at room temperature and pressure all the rest are solids. The vast majority of the known elements are metals. Nuclides are also characterized by its nuclear energy states (e.g. Metals-such as copper or gold-are good conductors of electricity and heat they can be pulled into wires because they are ductile they can be hammered or pressed into thin sheets or foils because they are malleable and most have a shiny appearance, so they are lustrous. In nuclear physics and nuclear chemistry, the various species of atoms whose nuclei contain particular numbers of protons and neutrons are called nuclides. The distinction between metals and nonmetals is one of the most fundamental in chemistry. Gold-colored lements that lie along the diagonal line exhibit properties intermediate between metals and nonmetals they are called semimetals. It is the arrangement of these electron orbits that is reflected in the different blocks of the periodic table.\) divides the elements into metals (in blue, below and to the left of the line) and nonmetals (in bronze, above and to the right of the line). The underlying factor that largely dictates an element’s properties is the number of electrons that orbit in the shell furthest form the nucleus. 
We now know the reality is more sophisticated. Isotope: Atoms of the same element with the same number of protons but a different number of neutrons. Ion: An atom or molecule with an electric charge due to the loss or gain of electrons. In tribute to this musical analogy, the chemists who first noticed the pattern in the elements more than 150 years ago called it the law of octaves. GCSE Chemistry Key Words Halogens: The elements in Group 7 of the periodic table. As you hit the eighth note, something beautiful happens – a note hangs in the air that embodies something of the first. The notes flow out, resounding at a higher and higher pitch as your hand moves to the right. Lithium, for example, with an atomic number of 3, is a reactive metal - just like sodium (element number 11) and potassium (number 19).Ī good way to think about it is to imagine running your fingers over the keys of a piano. As you keep adding protons, you find that similar properties recur in every 8th element. If you have one proton, that is hydrogen. It is the number of protons in an atom’s nucleus, known as its atomic number, that determines which element it represents. The periodic table captures a subtle pattern that runs through the chemical elements, the fundamental building blocks of everything around us: from the aluminium in bike frames to the xenon gas in glowing shop signs.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
